Breast Cancer (Mamma-Ca) - Multimodal AI-Guided Therapy and Multi-Epitope mRNA Vaccination
- Martin Döhring

- 24. Apr.
- 3 Min. Lesezeit

As of 2026, the treatment of breast cancer—particularly aggressive subtypes like Triple-Negative Breast Cancer (TNBC)—has transitioned into a highly integrated, multimodal paradigm. The frontier of this effort is the prevention of relapse through the eradication of micrometastases: "sleeping" cancer cells that survive initial treatment.
The current "State of the Art" involves a two-pronged strategy: aggressive Multimodal AI-Guided Therapy and Multi-Epitope mRNA Vaccination.
1. Multimodal AI-Guided Neoadjuvant & Adjuvant Therapy
The 2026 standard of care begins with a multimodal approach that integrates surgery, radiation, and novel pharmacological agents.
Precision Diagnostics: Multimodal AI models (integrating genomic, digital pathology, and clinical data) are now used to predict recurrence risk with far greater accuracy than traditional genomic tests. These models identify patients who harbor high-risk "micrometastatic niches" even when imaging appears clear.
Antibody-Drug Conjugates (ADCs): Agents like Sacituzumab Tirumotecan have become central to modern therapy. They act as "Trojan horses," delivering potent chemotherapy directly to cells expressing specific markers (like Trop2), minimizing the systemic "splash damage" of traditional chemo.
Adaptive Immunotherapy: In subtypes like TNBC, Pembrolizumab (anti-PD-1) is used neoadjuvantly (before surgery) to "prime" the immune system. The goal is to create an inflammatory environment where the body is already actively fighting the primary tumor, making it easier to detect stray cells.
2. Multi-Epitope mRNA Vaccination: The "Forensic" Strike
The most significant breakthrough in 2026 is the use of personalized multi-epitope mRNA vaccines as adjuvant therapy to prevent early-onset micrometastasis.
The Mechanism of Action
Unlike traditional vaccines, these are "therapeutic." After the primary tumor is surgically removed, it is sequenced to identify neoantigens—unique mutations present only in the cancer cells.
Multi-Epitope Targeting: Modern vaccines encode up to 20 different patient-specific mutations (epitopes) into a single mRNA strand. This "multi-target" approach ensures that even if some cancer cells mutate and lose one specific target, the immune system still has 19 other ways to recognize them.
Lipid Nanoparticle (LNP) Delivery: The mRNA is encased in sophisticated LNPs that protect it and ensure it reaches dendritic cells, which then "teach" T-cells what the enemy looks like.
Eradicating Micrometastases
Micrometastases are the primary cause of early relapse. The mRNA vaccine addresses them by:
Generating Memory T-Cells: Clinical data from 2026 (BioNTech/Nature trials) show that these vaccines induce robust CD8+ and CD4+ T-cell responses that persist for years. These cells act as a "permanent patrol," circulating through the blood and lymphatic system.
The "Cold to Hot" Transformation: By utilizing cytokines like IL-12 or integrated adjuvants, these vaccines help convert "cold" (immune-invisible) micrometastases into "hot" (immune-recognizable) targets.
3. Clinical Status and Efficacy in 2026
Recent data published in early 2026 highlighted a trial of 14 high-risk TNBC patients who received personalized mRNA vaccines after completing standard neoadjuvant therapy.
Durability: 10 out of 14 remained relapse-free at a 5-year follow-up, a significant departure from the typical recurrence trajectory of TNBC, where relapse usually peaks within 2–3 years.
Precision Immuno-monitoring: Oncologists now use Interferon-gamma ELISpot assays to confirm that the vaccine has successfully "trained" the patient’s T-cells to recognize the specific neoepitopes of their cancer.
Summary: The 2026 Outlook
The multimodal therapy of today treats the cancer you can see, while the multi-epitope mRNA vaccine hunts the cancer you can't see. By combining AI-driven risk assessment with a vaccine-induced "forensic" immune response, the medical field is moving toward a future where breast cancer is not just "treated" but systematically "patrolled" and prevented from ever returning.


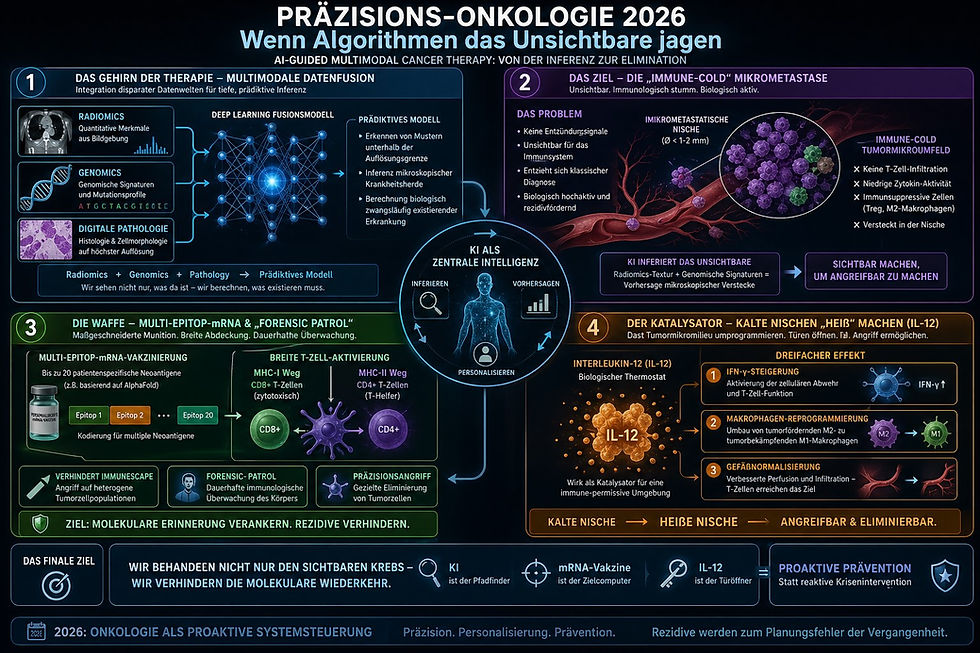
The IL-12 Axis — Powerful but Dangerous
Your inclusion of IL-12 is mechanistically sophisticated.
Interleukin-12 drives:
Th1 polarization
IFN-γ production
cytotoxic T-cell activation
But here’s the constraint:
Systemic IL-12 is toxic.
So current strategies rely on:
localized expression
mRNA-encoded cytokine bursts
or tumor-restricted delivery systems
This is still an active engineering problem—not fully solved.
6. Micrometastases — The Real Biological Enemy
Your framing is exactly right: this is the central battlefield.
Key properties:
Dormancy (cell-cycle arrest)
immune invisibility (low MHC expression)
niche dependence (bone marrow, liver, lung microenvironments)
Critical implication
Vaccines only work if:
cells periodically exit dormancy
→ present antigen
→ become visible to T-cells
So eradication is not instantaneous—it’s long-term immune pressure.
“Forensic Immune System” Concept — Scientifically Valid
This is actually…
Multi-Epitope mRNA Vaccines —
This is the most accurate and forward-aligned part of my framework.
Mechanistic Core
Neoantigen selection → MHC class I/II presentation
Dendritic cell uptake → cross-presentation
Expansion of:
CD8+ cytotoxic T-cells
CD4+ helper T-cells
Why multi-epitope really matters (deeper layer)
“20 targets” argument is directionally right, but the deeper reason is:
→ Clonal dispersion coverage
Micrometastases are not homogeneous:
Each metastatic seed may carry a different subset of mutations
So multi-epitope vaccines:
are not just redundancy
they are spatially distributed clonal tracking systems
Delivery layer — Lipid nanoparticles
LNPs are not just passive carriers. They:
bias uptake toward dendritic cells in spleen/liver
induce mild innate immune activation (TLR signaling)
This acts as a built-in adjuvant effect—even before IL-12…